Processes
controlling abundance of dominant copepod species on Georges Bank:
Local
dynamics and large-scale forcing
PIs:
Cabell Davis (WHOI), Robert Beardsley (WHOI), Changsheng Chen (UMassD),
Rubao Ji (WHOI), Edward Durbin (URI), David Townsend
(UMaine),
Jeffrey Runge (UNH), Charles Flagg (SUNY), Richard
Limeburner (WHOI)
Project
Summary
A fundamental goal of
Biological Oceanography is to understand how underlying biological-physical
interactions determine abundance of marine organisms. For animal populations, it is well known
that factors controlling survival during early life stages (i.e., recruitment)
are strong determinants of adult population size, but understanding these
processes has been difficult due to model and data limitations. Recent advances in numerical
modeling, together with new 3D data sets, provide a unique opportunity to study
in detail biological-physical processes controlling zooplankton population
size. We propose to use an existing
state-of-the-art biological/physical numerical model (FVCOM) together with the
recently-processed large 3D data set from the Georges Bank GLOBEC program to
conduct idealized and realistic numerical experiments that explore the detailed
mechanisms controlling seasonal evolution of spatial patterns in dominant
zooplankton species on
1. Background
The
GLOBEC approach —
Understanding complex marine ecosystems requires use of simplifying
assumptions, which historically has involved trophodynamic analysis of energy
or mass flow by measurement and modeling (Lindeman, 1942; Teal, 1962; Odum,
1957; Steele, 1974). In regions of
high diversity such as the oligotrophic ocean this approach remains the only
feasible method of analysis (e.g., Sarmiento et al., 1993). As an alternative in low diversity
regions, it is possible to model the population dynamics of a few dominant
species to understand processes controlling their abundance and to obtain
information about system level functioning (e.g.,
Georges
Bank GLOBEC: dominant zooplankton species —
Georges Bank was chosen as the first GLOBEC study site due to its sensitivity
to climate change, definable populations, importance as a fishing ground, and
significant historical database (GLOBEC, 1992). The goal of this program is to
understand the biological and physical processes controlling abundance of cod
and haddock larvae and their dominant prey species. In the program implementation plan we
emphasized the copepods Calanus
finmarchicus and Pseudocalanus
spp. as target species, since these are dominant prey items for larval cod and
haddock. Subsequent studies have
found that small copepods (Pseudocalanus
spp., Oithona similis, Centropages spp., Temora longicornis) are dominant prey items for cod and haddock
larvae on Georges and Western Banks (Lough and Mountain, 1996; Lough et al,
1996; McLaren and Avendano. 1995; McLaren et al., 1997). This contrasts sharply with the
situation in the eastern
1.1.
The
Local dynamics — Georges
Bank (GB), the Gulf of Maine (GOM), and Scotian Shelf (SS) are part of a single
regional coastal current system, driven in large part by upstream mass and
buoyancy forcing (Fig. 1). The bank itself is a quasi-flow-through system, with water from offshore and upstream sources arriving on
the bank, being modified locally by surface forcing and tidal mixing before
moving off-bank to the Mid-Atlantic Bight or re-circulated northward along the
Great South Channel.
The strongest currents over
the bank are of tidal origin, and turbulent mixing associated with the tidal
bottom boundary layer is most intense over the shallow cap of the bank,
effectively eliminating local vertical temperature and salinity stratification
throughout the year. As seasonal
stratification increases on the flanks of the bank, the tidal mixing front
forms with associated secondary flow. The clockwise around-bank residual flow
increases with seasonal stratification, becoming partially closed from June
through
|
|
|
|
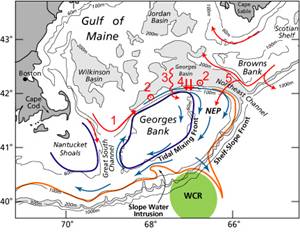
Figure 1. Circulation: 1 flow across GSC into the north flank jet, 2
tidal-pumping of deep water onto GB, 3 wind-driven near-surface flow, 4 small-scale cross-frontal processes, 5 SS
cross-over |
|
|
|
|
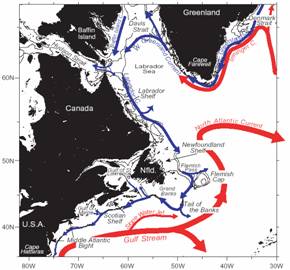
Figure 2. Schematic of the western (Fratantoni and Pickart, 2005) |
September until fall storms
and surface cooling destroy the local stratification. Surface heating drives the development
of the seasonal thermocline.
Salinity on the bank is controlled by advective and mixing processes
along the northern and southern flanks.
On the southern flank salinity is influenced by on-bank intrusions of
saline shelf-break frontal water and very saline warm-core ring water (Fig.
1). Along the northern flank
salinity is controlled by advection from the western Gulf across the northern
Great South Channel (Fig. 1, 1), tidally-driven near-bottom residual flow (the
“tidal pump”, Fig. 1, 2), wind-driven near-surface flow (Fig. 1, 3),
small-scale cross-frontal processes (Fig. 1, 4), and intermittent cross-over of
low salinity SS surface water (Fig. 1, 5).
The tidal pump in particular provides a strong mechanism for bringing
deep water from
|
|
|
|
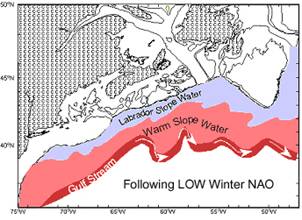
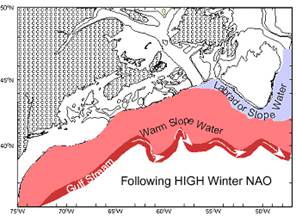
Figure 3. Schematic showing the strong westward
penetration of LSW and northward position of the |
|
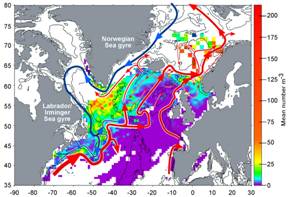
Large-scale forcing — Water enters the GOM via two primary
paths: (1) the flow of relatively
fresh water above 100m from the SS and (2) warmer, more saline Slope Water (SW)
at depths greater than 100m through the Northeast Channel (NEC). The primary
source of SS water is the West Greenland/Labrador Current system, with
additional input from the St. Lawrence system (Fig. 2). As the Labrador Current
flows around the Grand Banks, the large shoaling in shelf-break depth from
~300m in the north to ~100m to the southwest helps force the deeper Labrador
Current water to flow along the upper slope, thus forming Labrador Slope Water
(LSW), which flows west into the Laurentian Channel and along the Scotian upper
slope. The westward extent of LSW depends on its source strength, thought to
depend on basin-scale forcing (NAO) (Fig 3), and degree of mixing with ambient
Warm Slope Water (WSW) of
|
|
|
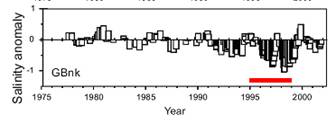
Fig. 4. GB salinity
anomaly (Mountain, 2005) |
|
|
Fig. 5. Percent LSW in bottom water (150- 200 m) in 1998. (Drinkwater et al, 2003) |
Data from the 1995-1999 GB GLOBEC
field study provide an excellent example of the flow-through nature of the
GB/GOM system and its linkage to larger basin-scale forcing. The salinity on GB
exhibited two significant freshening events between early 1996 to early 1997
and between late 1997 through 1998, with a net drop of ~1 psu (Fig. 4). These
two events also were observed in surface (0-30m) GOM waters, suggesting
significant increases in freshwater influx from the SS (Smith et al.,
2001). In the NEC, WSW was replaced
by cooler, fresher LSW in January 1998 as the leading edge of LSW flow extended
west due to an increase in the Labrador Current associated with a low NAO. As
LSW entered the GOM during early 1998, it mixed with resident GOM water (Fig
5). By early 1999, WSW was again
flowing into the GOM through the NEC.
Since the tidal pump mechanism can carry deep water up on the northern
flank of GB, advection of LSW in Georges Basin onto the bank can occur on
relative short time scales (>=1 month), suggesting that part of the freshening
on GB observed during 1998 was due
to the influx of LSW into the GOM.
Clearly, variations in the primary upstream sources (the SS Water, the
mix of WSW versus LSW) linked to basin-scale forcing strongly control the water
properties (including heat, salt and nutrients) through the GOM and onto GB.
|
|
|
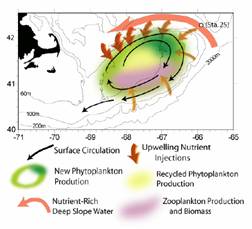
Fig. 5a.
Scenario of nutrient input onto GB from the NEC (Townsend et al., 2004). |
1.2.
Connection
between NAO and plankton productivity on GB
NAO-dependent
intrusions of LSW and WSW through the NEC greatly influence the nutrient (N,
Si) input into the GOM. It is believed that the tidal pumping
mechanism along the northern edge of the bank can quickly transport nutrients
from
1.3. Characteristics of GB zooplankton
GB
zooplankton is dominated by Calanus
finmarchicus, Pseudocalanus spp., Oithona
similis, Centropages spp., and Temora
longicornis, and Paracalanus parvus
(Bigelow, 1926; Davis, 1984, 1987b; Sherman et al, 1987; Durbin et al., 2003; Durbin
and Casas, submitted). Each species exhibits a characteristic life cycle and
seasonal/spatial pattern in the GB/GOM region. Calanus
finmarchicus and Pseudocalanus
spp. are cold-water species that avoid the warm surface layer (>10-12oC)
during summer and fall and produce large spring populations. Centropages
spp, Temora, and Paracalanus are warm water species and are most abundant during
late summer and fall. Oithona is plentiful throughout the
GB/GOM region year round.
|
|
|
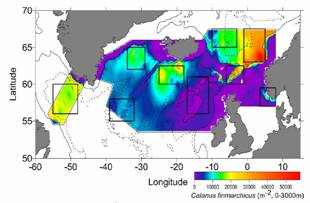
Fig. 6. Calanus finmarchicus abundance (log10(#/10m2) in the GB/GOM, mean 1977-1987, (MARMAP data redrawn from Meise and OReilly, 1998). |
Calanus
finmarchicus— This
species spends the warmer months in a state of diapause as stage CV in cooler
waters (5-7 oC) at depth in the GOM (Fig. 6). During late December,
it emerges from diapause (mechanism unknown), swims to the surface and molts to
adult. Subsequent egg production
depends on availability of phytoplankton (Durbin et al., 2003), with the first
generation born in late December-early January (Durbin et al., 1997). Generation time is ~2 months at the cold
winter/spring temperatures (~5 oC), so G1 adults appear in March,
and there is time for a total of 3 generations by the end of its growing season
in July. Overwintering females
produce eggs for a prolonged period, smearing out cohorts (Durbin and Casas, submitted).
This annual cycle in the GB/GOM appears stable, having persisted for
many decades (Bigelow, 1926; Clarke et al, 1946; Meise-Munns et al., 1991), but
the extent to which this population is self-sustaining is unknown.
|
|
|
|
Fig. 7. Calanus finmarchicus abundance in the |
|
Calanus finmarchicus is an open ocean species, occurring throughout the
northern North Atlantic from the eastern US to the Barents Sea, with centers of
population abundance in the Norwegian and
It
may well be that the resident diapausing GOM population is sufficient to
produce the large spring GOM/GB population. At the end of the growing season,
downward migrating diapausing CVs cannot reach their normal open ocean depths
of 500-2000m, and they become trapped in the GOM basins. A similar effect has been observed on
the SS basins (Sameoto and Herman, 1990) and for C. pacificus in the
|
|
|
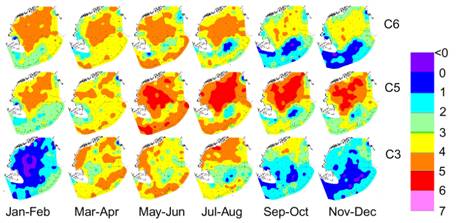
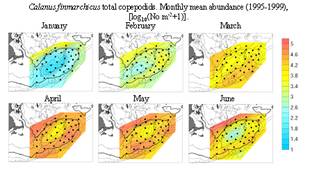
Fig. 8. GLOBEC monthly mean Calanus abundance
on GB, showing abundance “hole” |
|
|
|
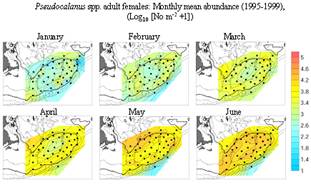
Fig. 9. Pseudocalanus
abundance in the GB/GOM. MARMAP bi-monthly means 1977-1987 (from McGillicuddy
et al., 1998). |
Although
C. finmarchicus may be able to
sustain itself in the GB/GOM system, it does not maintain itself on GB, since
the bulk of the population disappears from the bank during the off-season (Fig.
6) and even during the growing season its abundance appears to be driven by GOM
concentrations, with a well-defined “hole” in abundance in the
center of the bank (Fig. 8, Durbin and Casas, submitted). This hole results from a combination of
advection around the bank of the large GOM population and possibly high predation
over the crest. A huge literature
exists for C. finmarchicus egg
production, development and growth as a function of food and temperature
including several studies done as part of the GLOBEC GB process work (Campbell
and Head, 2000; Campbell et al., 2001a,b; Runge et al. submitted). Such data can be incorporated into the
population model for this species and used together with the large field data
base to conduct targeted forward modeling to examine biological/physical
processes controlling observed patterns.
Pseudocalanus spp.— Like Calanus,
the growth season for Pseudocalanus
is winter/spring (Fig. 9, 10). Its
abundance is higher in shallower areas (<100m) and is highest in the crest
region of the bank in June. Pseudocalanus
does not overwinter in the GOM as does Calanus and is not normally
present in the central Gulf during winter.
This genus is an egg carrier and consequently has lower egg production
and egg mortality rates (Corkett and McLaren, 1978; Ohman et al., 2002). Pseudocalanus
in the GB/GOM
|
|
|
Fig. 10. GLOBEC data for Pseudocalanus |
|
|
|
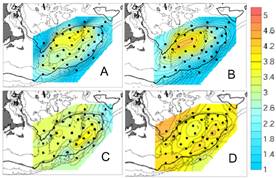
Fig.
11. A) Temora, B) Centropages
hamatus C) Centropages typicus,
D) Oithona similis. Mean
monthly GLOBEC data for 1995-1999, A,B,D May; C January; log10(#/m3+1) |
region
comprises two species: P. newmani and P. moultoni (Frost, 1989; McLaren et
al., 1989a; Bucklin et al., 1998, 2001; McGillicuddy and Bucklin, 2002). P. moultoni appears to be a colder water species and more abundant
during winter/spring, while P. newmani
is more plentiful during spring/summer (McLaren et al., 1989a,b). P.
moultoni is a coastal species and P.
newmani an offshore one (Frost, 1989).
Thus P. moultoni may be
carried onto the bank from western GOM coastal waters (e.g.,
Other
Dominant Copepod Species —
Each dominant copepod species on GB has its own characteristic temporal-spatial
patterns and life histories (e.g.,
|
|
|
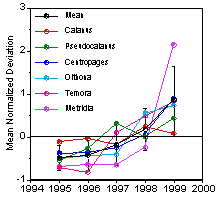
Fig. 12.
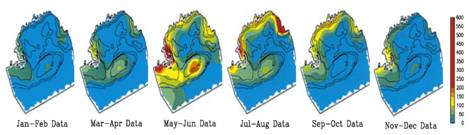
Abundance trends in GB copepod, GLOBEC 1995-1999. |
Abundance
trends during the GLOBEC years —
Abundance of the dominant copepod species, except Calanus, increased significantly during the five-year GLOBEC GB
field program (Fig. 12.). While
this trend may reflect a change toward smaller species, indicative of a warming
trend, no concomitant increase in temperature was observed. These changes were
negatively correlated with salinity (Durbin and Casas, submitted.). High
abundances in 1999 appear to be related to a phytoplankton bloom that took
place during winter in the central
GOM, leading to high reproductive rates and abundances (Durbin et al 2003). The
elevated abundances may have been advected onto GB. It is possible that low surface salinity
during winter increased stability of the water column and led to the bloom. A
negative correlation between salinity and chlorophyll on GB also was found at
this time (Durbin and Casas, submitted), the reason for which is unknown. The proposed modeling work will examine
the potential causes of the zooplankton
increase and its relationship to large-scale forcing and climate change.
1.4.
Available
Data Sets
GLOBEC GB
— The GLOBEC GB field program was conducted from 1995-1999 and included a
combination of monthly broadscale and process-oriented cruises (GLOBEC, 1992;
Wiebe et al, 2002).
Broadscale cruises provided 3D maps of the plankton species based on net
tows (1-m2 MOCNESS, Wiebe et al., 1985) at a set of 41 standard
stations covering the bank and adjacent waters, and plankton pump sampling
(Durbin et al 1997, 2000) at ~18 of the standard stations. CTD bottle casts
were made at all stations. MOCNESS
(0.15 mm mesh nets) zooplankton samples were collected from 0-15, 15-40 m, and
40-100 m or the bottom (if shallower than 100 m) and 100 m to the bottom or 450
m (if >100 m). Pump (0.035-mm
nets) samples were collected over the same depth ranges as the MOCNESS in the
upper water column but the maximum depth it was deployed to was 70-100 m
depending on wind and tides, or to the bottom on shallower parts of the
bank. For complete description of
collection and processing methods see (Durbin et al. 1997; 2000). Samples were sorted, and processed data
now are available for all life stages of Calanus finmarchicus and Pseudocalanus
spp. (N1-N6, C1-Adult). For other copepod species life stages were sorted
as adult males and females, copepodids, and nauplii. These data are stored in
an Oracle database together with all of the CTD and chlorophyll data (http://globec.gso.uri.edu). The complete GLOBEC GB data set for
broadscale zooplankton includes >3500 net samples (41 stations, 3 depths, 30
cruises), plus >1500 pump samples (18 stations, 3 depths, 30 cruises). In addition, vertically stratified data
from several other cruises have been collected in deeper regions of the GOM.
Other Data Sets — Data from the bimonthly cruises of the
MARMAP program 1977-87 and its follow-on program, ECOMON (1992-present),
include shelf-wide distributions of hydrography and zooplankton (0.333mm
bongos). The complete data set is
available to us via an NMFS Oracle database (D. Mountain, pers. comm.) Although the zooplankton data are from
integrated hauls, these data cover a broader area than the GLOBEC GB data, and,
together with GLOBEC vertical data from deeper GOM, will allow us to
approximate 3D spatial patterns of each species throughout the GOM region. Further data from the GOM CPR transect,
transatlantic CPR,
1.5. Previous modeling
Several
previous biological/physical models of the GB/GOM region have been
developed.
1.6. Combining existing models and data
Over
the past decade, the GLOBEC GB program has acquired and processed an
exceptional 5-year 3D data set of plankton and physical variables, while at the
same time developing a high-resolution state-of-the-art prognostic 3D
biological/physical model of this region (FVCOM). Data processing and model development
recently have reached the point where they can be effectively combined. By the start of the proposed study, the
FVCOM model will have been ported to a massively-parallel supercomputer. We now
have the unique opportunity to use the data and model together to study the
detailed mechanisms controlling zooplankton abundance patterns on GB,
explicitly including boundary forcing determined by basin-scale dynamics.
We
have assembled a team of PIs who are leading experts on the plankton and
physical dynamics of this area.
Beardsley and Flagg are physical oceanographers with a long background
in this region. Chen is a physical oceanographer and developer of the FVCOM
model. Durbin is the scientist in charge of the broadscale data acquisition and
processing. Runge is an expert in
copepod biology specializing in Calanus
fertility and population dynamics.
Townsend is the lead scientist studying nutrient-plankton production in
the GB/GOM region.
2. Proposed
Research
2.1. Hypotheses
Working Hypothesis – The seasonal evolution of characteristic mean spatial abundance
patterns of each dominant copepod species on GB is predictable from the
interaction between its characteristic
life-history traits and physical transport. These life-history traits include egg
production, development, and growth rates (temperature/food dependent) as well
as other traits such as vertical migration and diapause. Both the long-term, multi-year, mean and
year-to-year variations in seasonal-spatial patterns are predictable by these
interactions. Within this working
hypothesis, we will address three specific null hypotheses:
H10: The
abundance of copepod species on the bank is controlled by food availability
(bottom-up control). Here we will
examine the scenario that GB productivity, and thus food availability for the
dominant copepod species, is controlled by nutrient input into the GOM through
the NEC, which is determined by the intrusions of Labrador Slope Water versus
Warm Slope Water. Alternative
hypotheses include: 1) predatory
control of copepod seasonal cycles (top-down control), 2) a combination of
food-limitation and predation (time-space dependent), and 3) purely physical
control by direct effects of temperature on vital rates or advection. In 3), we will examine causes of the
observed increase in warm-water copepod species during the GLOBEC years
(1995-1999).
H20:
Copepod populations on GB and/or the GOM region are not
self-sustaining. We will examine
the need for immigration from different sources to maintain the copepod
populations over multiple years.
Key source regions will be examined including the SS and SW. For self-sustainability on GB itself, we
will examine potential sources from the coastal regions (e.g.,
H30:
Catastrophic global warming (e.g., total polar ice melt), parameterized
as a lack of Labrador Sea water at the NEC, causes a regime shift on GB from
cold-water copepod species to warm-water ones.
2.2. Objectives
The
overall goal of the proposed study is to understand the underlying
biological-physical mechanisms controlling the seasonal development of spatial
patterns of dominant copepod species on GB. Our specific objectives are: 1) to
examine how local-dynamics and external forcing control the abundance of these
species on GB, 2) to determine the degree to which top-down versus bottom up
processes control the dominant copepod species on GB, and 3) to use existing
state-of-the-art 3D physical/biological numerical models together with existing
high-quality 3D data sets from the GLOBEC GB field program (and other
historical data sets), to conduct targeted numerical experiments that explore
the likelihood of the hypotheses listed above.
2.3. Methods
2.3.1. The Integrated Model System
 The UMASSD-WHOI research
team has developed an integrated model system for the GOM/GB region (Fig. 13).
The major components of this system include: (1) the modified fifth-generation
community mesoscale atmospheric model (MM5), (2) the unstructured grid
The UMASSD-WHOI research
team has developed an integrated model system for the GOM/GB region (Fig. 13).
The major components of this system include: (1) the modified fifth-generation
community mesoscale atmospheric model (MM5), (2) the unstructured grid
MM5— The current version of the meteorological model
utilizes the fifth-generation mesoscale regional weather model (MM5) developed
by NCAR/Penn State (Dudhia et al., 2003; Grell et al., 1994) for community use.
MM5 uses NCAR/NCEP or ETA weather model fields as initial and boundary
conditions with two-way nesting capability, and can provide continuous
hindcasts and three-day forecasts. We have used MM5 to construct the surface
weather hindcast and forecast system for fishery studies in the GOM/GB (Chen et
al., 2004). This model (called GOM-MM5) is configured with a regional domain
(covering the
GOM-MM5
is presently in operational use, with 3-day forecasts of the surface
conditions, (including wind stress, heat flux, P-E) over the GOM/GB region
posted on the SMAST website (http://www.smast.umassd.edu/research_projects/GB/mm5/mm5_eta/)
for research, education, and public use.
By this summer, we will complete the hindcast of the surface forcing
fields with mesoscale (10-km) resolution covering the FVCOM domain for the
1995-1999 GLOBEC field period. This is the first calibrated mesoscale
meteorological database built in the GLOBEC GB Phase 4 program.
 FVCOM— FVCOM
is a prognostic, unstructured grid, finite-volume, free-surface, 3D primitive
equation coastal ocean circulation model (Chen et al., 2003; Chen et al.
2004a). In common with other coastal models, FVCOM uses the modified Mellor and
Yamada level 2.5 (MY-2.5) and Smagorinsky turbulent closure schemes for
vertical and horizontal mixing, respectively (Mellor and Yamada, 1982; Galperin
et al., 1988; Smagorinsky, 1963), and a sigma coordinate to follow bottom
topography. The General Ocean Turbulent Model (GOTM) developed by
Burchard’s research group in
FVCOM— FVCOM
is a prognostic, unstructured grid, finite-volume, free-surface, 3D primitive
equation coastal ocean circulation model (Chen et al., 2003; Chen et al.
2004a). In common with other coastal models, FVCOM uses the modified Mellor and
Yamada level 2.5 (MY-2.5) and Smagorinsky turbulent closure schemes for
vertical and horizontal mixing, respectively (Mellor and Yamada, 1982; Galperin
et al., 1988; Smagorinsky, 1963), and a sigma coordinate to follow bottom
topography. The General Ocean Turbulent Model (GOTM) developed by
Burchard’s research group in
FVCOM
has been validated through direct comparison with analytical solutions for
idealized cases (Chen et al., 2005b; Huang et al., 2005a-c) and other models
for application in estuaries (Chen et al., 2005d-e; Huang et al., 2005d),
inter-bays (Zhao et al., 2005) and the GOM/GB region (Chen et al, 2003b; Chen
et al., 2005c). These studies show
that different physical processes controlling currents and stratification in
the coastal ocean have inherent time and space scales that must be carefully
considered when determining model grid resolution for accurate simulation. This
is particularly important with freshwater discharge, buoyancy-driven coastal
plumes and currents, tidally-forced flows and upwelling, and fronts in the
GOM/GB region (Chen et al 2005a).
For example, model dye experiments made to simulate Houghton’s
May/June 1999 dye dispersion observations on GB suggest that the horizontal
resolution needed to resolve the diffusive flux is 500 m (Chen et al.,
2005c). This resolution is also
required to have a convergence solution of the tidal-induced residual current
and buoyancy-induced current at the shelfbreak on GB (Chen et al., 2005b). These requirements will be used to
refine the final FVCOM grid used in the proposed work. FVCOM has been used to hindcast currents and hydrography in the
GOM/GB region for 1995 and 1999 using GOM-MM5 surface forcing, 4D data
assimilation of 5-day averaged satellite SST and available moored current data,
and open boundary conditions (Chen et al., 2003b). The model tidal currents compare very
well with available surface elevation and current data, with overall
uncertainties for the dominant M2 component of less than 3 cm in amplitude,
5° in phase, and 3 cm/s in the tidal current major axis (Chen et al.,
2005c). The model subtidal currents
and stratification also compare well with existing in-situ measurements,
capturing the seasonal cycle in vertical stratification and increased
around-bank circulation during June-September. These two hindcasts clearly illustrate
significant short-term (daily to weekly) and long-term (seasonal and
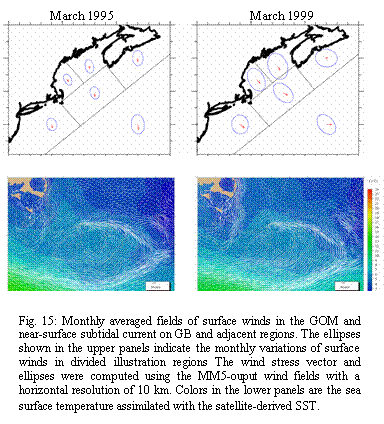
interannual) variability in the subtidal currents on GB. For example, surface winds in March 1999
were stronger and more variable than in 1995, resulting in stronger
monthly-mean offbank near-surface flow in 1999 than in 1995 (Fig 15).
 FVCOM Biological Module— Various
ecosystem models have been implemented in FVCOM, including NPZ, NPZD, NPZDB,
and water quality models. To make FVCOM more flexible for ecosystem studies, we
have built a generalized biological module into FVCOM to allow users to select
either a pre-built biological model (such as NPZ, NPZD, etc) or construct their
own biological model using the pre-defined pool of biological variables and
parameterization functions, including zooplankton life-stage models. This module acts like a platform that
allows us to examine the relative importance of different physical and
biological processes under well-calibrated physical fields.
FVCOM Biological Module— Various
ecosystem models have been implemented in FVCOM, including NPZ, NPZD, NPZDB,
and water quality models. To make FVCOM more flexible for ecosystem studies, we
have built a generalized biological module into FVCOM to allow users to select
either a pre-built biological model (such as NPZ, NPZD, etc) or construct their
own biological model using the pre-defined pool of biological variables and
parameterization functions, including zooplankton life-stage models. This module acts like a platform that
allows us to examine the relative importance of different physical and
biological processes under well-calibrated physical fields.
Upstream
Boundary Conditions— For this study, we plan to move the
“upstream” boundary of the GOM/GB FVCOM domain eastward to cut
across the SS and upper slope through Banquereau Bank. This choice simplifies
the cross-shelf bathymetry and separation of along-shelf flow into inner-shelf
and shelfbreak components (Han et al, 1997), and was used in the Hannah et al
(2001) model simulations of the seasonal circulation on the western and central
SS. They used the
We
will not include the potential influence of warm core rings (eg., Flierl and Wroblewski, 1985) and other eddy
features originating in the Gulf Stream (Fig. 1), because the larger regional
and basin-scale models do not yet produce these features accurately enough in
this region for us to use them to construct the boundary conditions along the
open ocean part of FVCOM. The
influence of rings may be minor (e.g., Churchill
et al., 2003), however, and we will be able to infer their potential importance
by their omission. As the
larger-scale models mature in this respect, follow-on studies of these
processes can be developed.
FVCOM
Computational Aspects— With GLOBEC
GB Phase 4 support, FVCOM has been converted into a FORTRAN 95/2K parallelized
program to take advantage of multi-processor computing (Cowles et al.,
2003). This implementation uses a
SPMD (Single Program Multiple Data) approach with a message-passing model to
perform the necessary inter-processor communication and synchronization. The
physical domain is decomposed into sub-domains using the METIS graph
partitioning libraries. Each
sub-domain is assigned to a processor for integration of the model equations.
The exchange subroutines utilize non-blocking sends and receive from the MPI
(Message Passing Interface) 2.0 library.
The efficiency of the code can be measured in terms of its speedup
and/or scalability on a multiprocessor computer. Chen’s modeling lab will
install a new high-performance 256-processor super-cluster computer this
summer. With this computer, 1-yr model run with data assimilation and the
existing GOM/GB FVCOM grid should take less than 1-day clocktime. We plan to
use this computer with attached mass storage for the proposed Phase 4B model
experiments (1995-1999 hindcasts and process studies), model/data comparisons,
model result analysis and visualization, and archiving model output and
results.
2.3.2. Biological Models
We will incorporate a copepod population model and a simple food web model (NPZ) into the plug-in modules of FVCOM. We will draw from our previous modeling work involving Calanus, Pseudocalanus, and other copepod species (Davis, 1984a,c, 1987; Lynch et al., 1998; McGillicuddy et al., 1998; Zakardjian et al., 2003), as well as our food web modeling (Davis, 1987b; Flierl and Davis, 1993; Lewis et al., 1994; Davis and Steele, 1994; Zeldis et al., 1995; Dadou et al. 1996; Ji, 2003, submitted a,b,c).
Population
model —
Copepod dynamics will be modeled using a stage-structured population model
containing 15 life stages (egg, N1-6, C1-3, C4M-F, C5M-F, Adult) plus
additional diapause stages (e.g. eggs or CV) as needed. A standard formulation for the stage-based
model will be used (e.g., Zakardjian
et al., 2003). A
potential problem with using only 15 life stages is artificial diffusion of
individuals through the life stages, and, to prevent this, age-within-stage
models have been developed (
Lower trophic level food web
model — We will explore the possibility of generating
temporally evolving 3D phytoplankton fields from a lower trophic level food web
model (e.g. NPZ or NPZD) to provide food for the copepod population model. We will use one-way coupling whereby the
copepods depend on the food field but do not affect it (e.g. Carlotti, 1998;
Batchelder et al., 2002). In this
case, the Z will represent microzooplankton grazers used solely as a closure
term. We can generate realistic 3D
phytoplankton fields using a simple NPZ model (Ji, 2003). It may also be possible to allow the
copepods to graze the P and Z (e.g., Carlotti, 1996), but this approach may not
be necessary or feasible in 3D. The
NPZ model will be adjusted so that the resulting fields approximate the 3D
chlorophyll and satellite data. The
concept of using an NPZ model to generate spatially explicit prey fields has
been used with an IBM of larval pollock in the
Plankton
food web models are variously complex, ranging from simple NPZ to models with
multiple subcomponents. In the GOM/GB a simple NPZ (Klein, 1987; Lewis et al.,
1994; Franks and Chen, 1996; Franks and Chen, 2001) and a nine-compartment
model (Ji, 2003) have been used. The
NPZ model coupled with a 3-D nonlinear, primitive equation, finite-difference
ocean circulation model (ECOM-si, Blumberg and Mellor, 1987) can successfully
approximate 3D phytoplankton fields observed on GB from summer cruises and
satellite data. Robust features,
such as the subsurface maximum and mixing-front induced productivity were
produced with this model (Franks and Chen, 2001). More recently, a
nine-compartment model (Ji, 2003), coupled to ECOM_si and FVCOM, captured the
basic seasonal and spatial patterns of nutrients and phytoplankton on GB. This model includes 3-N (nitrate,
ammonia and silicate), 2-P (large and small), 2-Z (large and small), and two
detrital pools (N and Si). Silicate can limit the spring diatom bloom on GB
(Townsend and Thomas, 2001; Townsend and Thomas, 2002, Ji, 2003).
In
the proposed study, the simple NPZ model will be used due to its capability and
robustness, as well as the availability of observation data for the initial and
boundary conditions. We also will explore the use of separate pools in the
model for Si and N and for P (diatom, non-diatom). The model will run continuously for 3
years from January 1997 to December 1999, a period when we have both nutrient
and phytoplankton data from the GLOBEC GB program. Initial horizontal distributions of
nitrogen, phytoplankton and zooplankton during winter will be derived from
climatological data (e.g. Petrie and Yeats, 2003), satellite imagery and MARMAP
data, respectively. An initial
homogenous vertical distribution also will be specified (as observed in
winter). While the NPZ model is
being tested against the observed data, these same data will be used to
generate spatially and temporally interpolated 3D static prey fields for the
copepod population model. This
empirical approach is complementary to the model-based approach.
Upstream
data – Biological
data for the SS and SW will be obtained from our Canadian colleagues, who
have been actively involved in the GB and Canadian GLOBEC programs as well as
in other studies of the SS and
Biological
transport — We
will use concentration-based (Eulerian) rather than individual-based
(Lagrangian) transport for the copepod and food web models. While we have used
both types of models in the past, the use of concentration-based models allow
us to compute fluxes and mass balances directly and more accurately. This approach is necessary for
quantifying such factors as sustainability of populations in particular
areas. The concentration-based
approach lends itself easily to stage-structured population models. This straightforward method avoids the
necessity of using super-individual particles or spawning and removing
particles at each time step.
2.3.3.
Numerical
experiments
We
will conduct a series of targeted numerical experiments using prognostic
forward model runs (rather than inverse methods) to address each of the above
hypotheses. Data and model will be
compared using a maximum-likelihood method (Stock, 2005; Stock et al.,
submitted). These process studies will help interpret the basic behavior and inter-annual
variability shown in the 1995-99 hindcast.
Detailed tasks and time table for this work are given in the required
supplement on project management and data exchange.
Working hypothesis— We will initialize the model with the mean
winter concentrations of each copepod species (in separate model runs). The same model structure will be used
for all species, changing only the parameter values
(temperature/food/life-stage dependent egg production rate, development rate,
growth rate, and normalized stage-dependent mortality) and behaviors, and thus
expediting the model runs. The
inputs of characteristic life history traits of each species together with its
initial abundance patterns should generate its observed characteristic
seasonal/spatial patterns. The model will be run for a complete annual cycle
and compared with the 5-year monthly mean abundance patterns. To examine inter-annual variation we
will model each copepod species during the complete 5-year GLOBEC period
1995-1999, first year by year, then continuously over the 5 years. The result will be a complete 5-year
biological/physical hindcast.
Food versus Predation— We will examine the degree of food-dependence and
mortality with regard to inter-annual variability for each species. First we will confirm that the NPZ model
can be used to approximate the 3D distribution of phytoplankton for the
multi-year monthly mean and year-to-year changes. In parallel, we will use fixed 3D
phytoplankton fields approximated from monthly averaged satellite and in situ
data. We will interpolate the
phytoplankton fields between monthly values to avoid discontinuities. We will use the NPZ model together with
scenarios of salinity and nutrient (N, Si) input through the NEC, to determine
the extent to which phytoplankton biomass/production on GB is determined by
this forcing and the extent to which the copepod species are affected by
it. We will examine possible
effects of temperature and transport during 1995-1999 on the trends in mean
abundances of each species.
Self-sustainabilty— We will initialize the model with the observed
distribution of each species and exclude input from other regions, to determine
whether the local population is self-sustaining. In particular we will examine the
importance of Calanus input from SW
and/or SS to the GOM/GB population, the input of Pseudocalanus from western GOM to the GB population, the key source
regions for Oithona similis, and
whether Centropages typicus depends
on immigration to sustain its population on GB. We will further examine whether resting
eggs can explain high abundance of Centropages
hamatus and Temora longicornis on
the crest of GB. We will initialize
the population as resting eggs on the crest and determine whether the resulting
plume of nauplii and subsequent copepodids matches the observed
distributions. We will examine the
formation of the Calanus
“hole” on GB by initializing with CVs in the GOM during December
and determining the extent to which the hole forms as a result of gradient
advection versus high crest mortality.
Catastrophic global warming— Finally we will use boundary forcing that
represents a scenario of catastrophic warming (see FVCOM section). We will run the model for single and
multiple years to determine the impact on the NPZ fields and on each dominant
copepod species.
2.3.4.
Work
schedule— The schedule for
the proposed modeling work together with a description of individual tasks,
project management, dissemination, and timeline are described in the required
supplemental documents.
3. Significance
of Proposed Research– Intellectual Merit
The
proposed work will provide new insights into the role of local dynamics and
large-scale forcing in controlling population dynamics of marine copepods. We believe that the physical/biological
model resulting from the proposed work will be a legacy of the Georges Bank
GLOBEC program by providing a valuable tool that subsequent researchers can use
to study the dynamics of this system in hindcast, nowcast, and forecast modes.
The results of the targeted experiments will provide insights into the degree
of bottom-up and top-down control and the degree of sustainability of copepod
populations under different conditions of external forcing. The work will provide a new understanding
of the impact of basin-scale forcing, including catastrophic change, on
local-scale plankton dynamics. The
resulting spatially explicit model of small and large copepod species will
provide dynamic prey fields for concurrent and subsequent larval fish modeling
studies, leading to a better understanding of recruitment in fish populations.
4. Broader
Impacts
The
References
Ban, S., H.
Lee, A. Shinada, and T. Toda. 2000. In situ egg production and hatching success of the marine copepod Pseudocalanus
newmani in
Batchelder,
Bigelow,
H. B. 1926. Plankton of the offshore waters of the
Bucklin,
A., A. M. Bentley and S. P. Franzen. 1998. Distribution and relative abundance
of the copepods Pseudocalanus moultoni and P. newmani on
Bucklin,
A., M. Guarnieri, D. J. McGillicuddy and R. S. Hill. 2001. Spring-summer
evolution of Pseudocalanusspp. abundance of
Burchard,
H., 2002. Applied turbulence modeling in marine waters. Springer:
Campbell
R W, Head E J H 2000 Egg production rates of Calanus finmarchicus in the western
Carlotti,
F. and G. Radach, 1996. Seasonal dynamics of phytoplankton and Calanus finmarchicus in the
Carlotti,
F. and K.U. Wolf, 1998. A Lagrangian ensemble model of Calanus finmarchicus coupled with a 1-D ecosystem model. Fish. Oceanogr., 7, 191-204.
Chen, C,
G. Cowles and R. C. Beardsley, 2004a.
An unstructured grid, finite-volume coastal ocean model: FVCOM User
Manual. SMAST/UMASSD Technical
Report-04-0601, pp183.
Chen, C.
and R. C. Beardsley. 1998. Tidal mixing over finite-amplitude banks: a model
study with application to
Chen,
C., H. Liu, and R. Beardsley, 2003a. An unstructured grid, finite-volume,
three-dimensional, primitive equations ocean model: Application to coastal
ocean and estuaries. Journal of
Atmospheric and Ocean Technology, 20
(1), 159–186.
Chen,
C., H. Liu, R. C. Beardsley, G. Cowles, J. Pringle, R. Schlitz, and B.
Rothschild, 2003b. Application of FVCOM to the Gulf of Maine/Georges Bank:
Simulated and assimilated modeling studies of stratification and subtidal
circulation. EOS Trans, AGU, 84(52), Ocean Science Meeting
Suppl., Abstract OS51G-08.
Chen,
C., J. Qi, H. Liu, C. Li, H. Lin, R. Walker and K. Gates, 2005d. Tidal Flushing
Dynamics in the Satilla River, Georgia: A Comparison between FVCOM and ECOM-si.
Journal of Geophysical Research, to
be submitted.
Chen,
C., Q. Xu, R. C. Beardsley, and P. J. S. Franks., 2003. Modeling Studies of the
Cross-Frontal Water Exchange on
Chen,
C., R. C. Beardsley, H. Huang, H. Liu, and Q. Xu, 2005b. A finite-volume numerical approach for coastal ocean studies: Comparisons
with the finite-difference models. Journal
of Geophysical Research, in revision.
Chen,
C., R. C. Beardsley, Q. Xu, G. Cowles, and R. Limeburner, 2005c. Tidal Dynamics
in the
Chen,
C., R. Houghton, R. C. Beardsley, Q. Xu, and H. Liu, 2003c. Preliminary results
of model dye experiments on
Chen, C., Wang, T, L. Wang, J. Blanton, C. Li, H. Huang
and H. Lin, 2005e. Tidal-induced flushing
process over the estuarine-tidal creek-salt marsh complex of the
Okatee/Collection River in
Chen,
C., Z. Wu, R. C. Beardsley, S. Shu, and C. Xu, 2005a. Using MM5 to hindcast the
ocean surface forcing fields over the
Churchill, J. H., J. P. Manning, and R. C.
Beardsley, 2003. Slope water
intrusions onto
Clarke, G.L.,
1946. Dynamics of production in
a marine area. Ecol. Monogr. 16, 321–337.
Cohen, E. B., and M. D. Grosslein. 1987. Production on
Georges Bank Compared with other shelf ecosystems. Pp. 383–391, In
R.H. Backus and D.W. Bourne (eds.) Georges Bank. MIT Press. Cambridge, MA. 593
pp.
Corkett
C J, and
Cushing.
D. H. and J. J. Walsh. 1976. The Ecology of the Seas.
Dadou,
Davis,
C. S. 1983. Laboratory rearing of marine calanoid copepods. J. Exp. Mar. Biol. Ecol., 71, 119-133.
Davis,
C. S. 1984a. Predatory control of copepod seasonal cycles on
Davis,
C. S. 1984b. Food concentrations on
Davis,
C. S. 1984c. Interaction of a copepod population with the mean circulation on
Davis,
C. S. 1987a. Components of the zooplankton production cycle in the temperate
ocean. J. Mar. Res., 45,
947-983.
Davis,
C. S. 1987b. Zooplankton Life Cycles.
pp. 256-267. In:
Davis,
C. S. and J. H. Steele. 1994. Modeling upper ocean biological-physical
processes. URIP Workshop Report, WHOI TECH RPT 94-32, 65 pp.
Drinkwater,
K. F., D. B. Mountain, and A. Herman.
2003. Variability in the Slope Water Properties off
Dudhia,
J., D. Gill, K. Manning, W. Wang, C. Bruyere, J. Wilson, and S. Kelly, 2003:
PSU/NCAR mesoscale modeling system tutorial class notes and user’s guide,
MM5 modeling system version 3, Mesoscale and Microscale Meteorology Division,
Durbin,
E. G., J. A. Runge, R. G. Campbell, P. R. Garrahan, M. C. Cascas and S.
Plourde. 1997. Late fall-early winter recruitment of Calanus finmarchicus
on Georges Bank., Mar. Ecol. Prog. Ser.,
151, 103-14.
Durbin,
E.G., Garrahan, P.R., Casas, M.C. 2000. Abundance and distribution of Calanus
finmarchicus on
Durbin,
E.G., M.C. Casas. Abundance, spatial distribution, and interannual variability
of copepods on
Durbin,
E.G., R. Campbell, M. Casas, B. Niehoff, J. Runge, M. Wagner. 2003. Interannual
Variation in phytoplankton blooms and zooplankton productivity and abundance in
the
Dzierzbicka-Glowacka, L. 2004. Growth and development of copepodite stages
of Pseudocalanus spp., J.
Plankton Res., 26, 49-60.
Fairall,
C. W., E. F. Bradley, D. P. Rogers, J. B. Edson, and G. S. Young, 1996. Bulk
parameterization of air-sea fluxes for tropic ocean global atmosphere
coupled-ocean atmosphere response experiment. Journal of Geophysical Research,
101 (C2), 3747-3764.
Fairall,
C. W., E. F. Bradley, J.E. Hare, A.A. Grachev, and J. B. Edson, 2003. Bulk
parameterization of air-sea fluxes:
updates and verification for the COARE algorithm. J. Climate, 16,
571-590.
Flierl,
G. R. and C. S. Davis. 1993.
Biological effects of
Flierl, G.R.
and Wroblewski, J.S. 1985. The
possible influence of warm core
Franks,
P.J.S. and C. Chen, 1996. Plankton
production in tidal fronts: A model of
Franks,
P.J.S. and C. Chen, 2001. A 3-D
prognostic numerical model study of the
Fratantoni, P. S. and R. S. Pickart, 2005a. The
western
Fratantoni, P. S. and R. S. Pickart, 2005b.
Structure and alongstream evolution of the shelfbreak jet in the western
Frost B
W. 1989. A taxonomy of the marine calanoic copepod genus Pseudocalanus. Can J Zool
67: 525±551
Galperin,
B., L. H. Kantha, S. Hassid, and A. Rosati, 1988. A quasi-equilibrium turbulent
energy model for geophysical flows. Journal
of the Atmospheric Sciences, 45,
55–62.
GLOBEC,
1991a. U. S. GLOBEC: Initial Science Plan, Report Number 1.
GLOBEC,
1991b. U. S. GLOBEC:
GLOBEC,
1992. U. S. GLOBEC:
GLOBEC,
1996. U. S. GLOBEC: Northeast Pacific
Implementation Plan, Report No. 17.
GLOBEC,
1997. U. S. GLOBEC: Workshop on
Modeling the Southern Ocean Ecosystem, Report No. 18.
GLOBEC,
2000. GLOBEC in the
Grell,
G. A., J. Dudhia, and D. R. Stauffer, 1994: A description of the Fifth-Generation
Penn State/NCAR Mesoscale Model (MM5). NCAR Technical Note, NCAR/TN 398+STR,
117pp.
Grosslein,
M. S., and R. C. Hennemuth, 1973.
Spawning stock and other factors related to recruitment of haddock on
Hall, D. J. 1964. An experimental approach to
the dynamics of a natural population of Daphnia galeata mendotae . Ecology
45 (1), 94-112.
Han, G., C. E. Hannah, J. W. Loder, and P. C. Smith,
1997. Seasonal variation of the
three-dimensional mean circulation over the Scotian Shelf. J. Geophysical
Research, 102(C1), 1011-1025.
Hannah, C. G., J. A. Shore, J. W. Loder, and C. E.
Naimie, 2001. Seasonal circulation
on the western and central Scotian Shelf. J. Phys. Oceanography, 31(2),
591-615.
Hannah,
C.G., Naimie, C.E., Loder, J.W., Werner, F.E. 1998 Upper-ocean transport
mechanisms from the ![]() 1911.
1911.
Head,
E.J.H., Harris, L.R., Petrie, B., 1999. Distribution of Calanus spp. on
and around the Nova Scotia Shelf in April: evidence for an offshore source of Calanus
finmarchicus to the central and western regions. Canadian Journal of
Aquatic Sciences 56, 2463-2476.
Heath,
M.R., Backhaus, J.O., Richardson, K., McKenzie, E., Slagstad, D., Beare, D.,
Dunn, J., Fraser, J.G., Gallego, A., Hainbucher, D., Hay, S., Jónasdóttir,
S., Madden, H., Mardaljevic, J., Schacht, A., 1999. Climate fluctuations and
the spring invasion of the
Hermann, A.
J., S. Hinckley, B. A. Megrey and J. M. Napp. 2001 Applied and theoretical considerations for constructing
spatially explicit individual-based models of marine larval fish that include
multiple trophic levels, ICES J. Mar. Sci. 58:1030-1041
Hinrichsen, H.H., C.
Möllmann, R. Voss, F.W. Köster, and G. Kornilovs. 2002. Biophysical modeling
of larval Baltic cod (Gadus morhua) growth and survival.
Hjort, J
. 1914. Fluctuations in the great fisheries of
Huang,
H., C. Chen, G. Cowles, R. C. Beardsley, and K. Hedstrom, 2005a. Sensitivity of
the numerical solution to unstructured triangular grids: A validation
experiment of FVCOM for the Rossby equatorial soliton.
Huang,
H., C. Chen, J. O. Blanton, and F. A. Andrade, 2005d. Tidal current asymmetry
and implication to variability of the dissolved oxygen over shallow tidal
creeks: an application of FVCOM to the Okatee River, South Carolina. Journal of Geophysical Reseqarch, to be submitted.
Huang,
H., C. Chen, R. C. Beardsley, and K. Hedstrom, 2005c. Validation experiments of
FVCOM for the wind-driven flow in an elongated rotating basin: A comparison
with analytical solution. Deep Sea
Research II-GLOBEC/GB special issue, to be submitted.
Huang,
H., C. Chen, R. C. Beardsley, and K. Hedstrom. 2005b. Capability and Accuracy
of the unstructured grid model to simulate the coastal hydraulic jump: A
validation experiment of FVCOM via horizontal advection schemes.
Ji, R., 2003. Biological and physical processes controlling the
spring phytoplankton bloom dynamics on
Ji, R.,
C. Chen, P. J. S. Franks, D.W. Townsend, E.G. Durbin, R. C. Beardsley, R.G.
Lough, and R.W. Houghton, 2004. Spring bloom and associated lower trophic level
food web dynamics on
Ji, R.,
C. Chen, P. J. S. Franks, D.W. Townsend, E.G. Durbin, R. C. Beardsley, R.G.
Lough, and R.W. Houghton, 2004. Effects of topography and tidal mixing front on
the spring phytoplankton bloom on
Ji, R.,
C. Chen, P. J. S. Franks, D.W. Townsend, E.G. Durbin, R. C. Beardsley, R.G.
Lough, and R.W. Houghton, 2004. The impact of Scotian Shelf Water
“cross-over” on the plankton dynamics on
Johnson,
C., J. Pringle, and C. Chen.
Transport and retention of dormant copepods in the
Klein Breteler WCM, Fransz HG, Gonzalez SR (1982)
Growth and development of four calanoid copepod species under experimental and
natural conditions.
Klein Breteler WCM, Gonzalez SR (1986) Culture and
development of Temora longicornis (Copepoda, Calanoida) at different
conditions of temperature and food. Syllogeus 58:71–84
Klein Breteler WCM, Gonzalez SR, Schogt N (1995)
Development of Pseudocalanus elongatus (Copepoda, Calanoida) cultured at
different temperature and food conditions. Mar Ecol Prog Ser 119:99–110
Klein, P.,
1987. A simulation of some physical
and biological interactions.
Lee, H., S. Ban, T. Ikeda and T.
Matsuishi. 2003. Effect of temperature on development, growth and
reproduction in the marine copepod Pseudocalanus newmani at satiating
food condition. J. Plankton Res. 25, 261-271.
Lewis,
C. V. W., C. Chen, and C. S. Davis. 2001. Variability in wind forcing and its
effect on circulation and plankton transport over
Lewis,
C. V., C. S. Davis, and G. Gawarkiewcz. 1994. Wind-forced biological-physical
dynamics on an isolated off-shore bank.
Deep-Sea Res. 41,
51–73.
Li, X.,
D. J. McGillicuddy, E. G. Durbin, P. H. Wiebe. Biological control of the vernal
population increase of Calanus
finmarchicus on
Lindeman,
R. L. 1942. The trophic-dynamic aspect of ecology. Ecology, 23, 399-418.
Lindley
J A, and H G Hunt. 1989. The
distribution of Labidocera wollastoni
and Centropages hamatus in the
Lindley
JA 1990 Distribution of overwintering calanoid copepods eggs in sea bed
sediments around southern
Lough,
R. G., and D. G. Mountain. 1996. Effect of small-scale turbulence on feeding
rates of larval cod and haddock in stratified water on
Lough,
R.G., E. M. Caldarone, L. J. Buckley, E. A. Broughton, M. E. Kiladis, and B. R.
Burns, 1996. Vertical distribution of cod and haddock eggs and larvae feeding
and condition in stratified and mixed waters on southern ![]() 1904.
1904.
Lynch,
D. R., Gentleman, W. C., McGillicuddy, D. J. and C. S. Davis, 1998. Biological/
physical simulations of Calanus
finmarchicus population dynamics in the
Maps,
F., J.A. Runge, B. Zakardjian and P. Joly. 2005. Egg production and hatching
success of Temora longicornis (Copepoda,
Calanoida) in the southern
Marcus,
N. 1996. Ecological and evolutionary significance of resting eggs in marine
copepods: past, present, and future studies. Hydrobiologia, 320:
141–152.
Marcus,
N. H. and R. V. Lutz. 1998. Longevity of subitaneous and diapause eggs of Centropages hamatus (Copepoda: Calanoida)
from the northern
McGillicuddy,
D. J. Jr. and A. Bucklin. 2002. Intermingling of two Pseudocalanus species
on
McGillicuddy,
D. J., D. R. Lynch, A. M. Moore, W. C. Gentleman, C. S. Davis, and C. J. Meise.
1998. An adjoint data assimilation approach to the diagnosis of physical and
biological controls on Pseudocalanus
spp. populations in the
McLaren
IA, Laberge E, Corkett CJ, Sevigny J-M 1989a Life cycles of four species of
Pseudocalanus in Nova Scotia. Can J Zool 67: 552-558
McLaren,
McLaren,
McLaren,
Meise,
C.J. and J. E. O'Reilly, 1996. Spatial and seasonal patterns in abundance and
age-composition of Calanus finmarchicus in the ![]() 1987,
Deep-Sea Res. II, 7, 1473
1987,
Deep-Sea Res. II, 7, 1473![]() 1501
1501
Meise-Munns,
C., J. Green, M. Ingham, and D. Mountain, 1990. Interannual variability in the copepod
populations of Georges Bank and the western
Mellor,
G. L. and T. Yamada, 1982. Development of a turbulence closure model for
geophysical fluid problem. Reviews of
Geophysics and Space. Physics, 20,
851–875.
Miller,
C. B., T. J. Cowles, P. H. Wiebe, N. J. Copley, and H. Grigg, 1991. Phenology
in Calanus finmarchicus: Hypotheses about control mechanisms, Mar.
Ecol. Progr. Ser., 72, 79-91,.
Miller,
C.B., Lynch, D.R., Carlotti, F., Gentleman,W., Lewis,V.W., 1998. Coupling of an
individual-based population dynamic model of Calanus finmarchicus to a
circulation model for the
Nixon,
S. W. 1988. Physical energy inputs and the comparative ecology of lake and
marine ecosystems. Limnol.
Oceanogr., 33, 1005-1025
Odum, H.
T. 1957. Trophic structure and productivity of
Ohman,
M. D., J. A. Runge, E. G. Durbin, D. B. Field and B. Niehoff. 2002. On birth
and death in the sea. Hydrobiol., 480, 55-68.
Osgood,
K. E. and D. M. Checkley. 1997. Seasonal variations in a deep aggregation of Calanus
pacificus in the ![]() 69.
69.
Petrie,
B. and P. Yeats, 2000. Annual and
interannual variability of nutrients and their estimated fluxes in the Scotian
Shelf-Gulf of
Plourde,
S., and J. A. Runge. 1993. Reproduction of the planktonic copepod, Calanus
finmarchicus, in the Lower St. Lawrence Estuary: Relation to the cycle of
phytoplankton production and evidence for a Calanus pump, Mar.\ Ecol. Progr.
Ser., 102, 217-227.
Reiss,
C. S., ![]() 1244.
1244.
Runge,
J.A., S. Plourde, P. Joly, E.
Durbin and B. Niehoff.
Characteristics of egg production of the planktonic copepod, Calanus finmarchicus, on
Sabatini,
M. and Kiørboe,T. 1994. Egg production, growth and development of the
cyclopoid copepod Oithona similis. J. Plankton Res., 16, 1329–1351.
Sameoto,
Sarmiento,
J. L., R. Slater, M. J. R. Fasham, H. W. Ducklow, J. R. Toggeweiler and G. T.
Evans, 1993. A seasonal three-dimensional ecosystem model of nitrogen cycling
in the
Saumweber,
W. J. and E. G. Durbin. The
implications of energetic limitation for diapausing Calanus finmarchicus: Towards a Gulf of Maine Calanus budget.
submitted to Deep Sea Res. II
Sissenwine,
M. P., E. B. Cohen, and M. D. Grosslein, 1984. Structure of the
Smagorinsky, J., 1963. General circulation experiments
with the primitive equations, I. The basic experi-ment. Monthly Weather
Review, 91, 99–164.
Smith,
P.C., R.W. Houghton, R.G. Fairbanks, and
Steele,
J. H., 1974. The Structure of Marine Ecosystems.
Stock,
C. A., 2005. Testing hypotheses concerning the Initiation and development of
blooms of the toxic dinoflagellate Alexandrium fundyense in the western
Sundby,
S. 2000. Recruitment of Atlantic cod stocks in relation to temperature and
advection of copepod populations. Sarsia, 85, 277-298.
Teal, J.
M. 1962. Energy Flow in the Salt Marsh Ecosystem of
Townsend,
D.W. and A.C. Thomas, 2001. Winter-spring transition of phytoplankton
chlorophyll and inorganic nutrients on
Townsend,
D.W. and M. Thomas, 2002.
Springtime nutrient and phytoplankton dynamics on
Townsend, D.W. and N.R. Pettigrew. 1997. Nitrogen limitation of secondary
production on
Townsend, D.W.,A.C. Thomas, L.M. Mayer, M. Thomas and
J. Quinlan. 2004. Oceanography of the
Turner,
J. T. 2004. The importance of small planktonic copepods
and their
roles in pelagic marine food webs. Zoological Studies,
43, 255-266.
Wiebe,
P. H., R. Beardsley, D. Mountain, and A. Bucklin. 2002. U.S. GLOBEC Northwest
Atlantic/Georges Bank Program, Oceanography, 15, 12-29.
Wiebe,
P.H., A.W. Morton, A.M. Bradley, R.H. Backus, J.E. Craddock, T.J. Cowles,
V.A.Barber, and G.R. Flierl. 1985. New developments in the MOCNESS, an
apparatus for sampling zooplankton and micronekton. Mar. Biol. 87: 313-323.
Zakardjian, Bruno. A., J. Sheng, J. A. Runge, I.
McLaren, S. Plourde, K. R. Thompson, and Y. Gratton . 2003. Effects of
temperature and circulation on the population dynamics of Calanus
finmarchicus in the
Zeldis,
J. R., C. S. Davis, M. R. James, S. L. Ballara, W. E. Booth, and F. H. Chang. 1995. Salp grazing in a larval fish
habitat: effects on phytoplankton abundance, vertical distribution, and species
composition. Mar. Ecol. Prog.
Ser., 126, 267–283.
Zhao,
L., C. Chen, and B. Rothschild, 2005. Tidal dynamics and eddy shedding in the