U.S. GLOBEC: R/V Oceanus Cruise OC334
to the Gulf of Maine
Acknowledgments
We would like to thank the captain and crew of the R/V Oceanus for a productive cruise; their professionalism was greatly appreciated. We also thank the US GLOBEC Georges Bank Program for supporting this cruise, with research support provided by NOAA. Support for the bio-optical oceanographic research conducted on this cruise was provided by an award from the ONR Young Investigator Program.
This report was prepared by Chuck Greene, Mark Benfield, Heidi Sosik, Peter Wiebe, Ann Bucklin, Louise McGarry, and Karen Fisher. The cruise was sponsored by the National Science Foundation and the National Oceanographic and Atmospheric Administration.
TABLE OF CONTENTS
INTRODUCTION3
CRUISE NARRATIVE4
INDIVIDUAL REPORTS7
1.0 Principal Project: Processes Regulating the Recruitment of Calanus finmarchicus from the Gulf of Maine to Georges Bank7
1.1 Introduction7
1.2 Methods8
1.2.1 Broad-scale survey8
1.2.1.1 Multi-frequency acoustics8
1.2.1.2 Video Plankton Recorder11
1.2.2 MOCNESS Zooplankton Samples11
1.3 Results11
1.3.1 Broad-scale survey11
1.3.1.1 Multi-frequency acoustics11
1.3.1.2 Video plankton recorder (VPR)15
1.3.2. MOCNESS Zooplankton Samples15
2.0 Ancillary Project: Spatio-Temporal Variability of Bio-Optical Properties in the Gulf of Maine 16
2.1. Introduction16
2.2. Methods16
2.3. Results17
CRUISE PARTICIPANTS18
Scientific Personnel18
Ship's Officers and Crew19
APPENDICES20
Appendix 1. Event Log21
Appendix 2. Acoustics Log23
Appendix 3. Cross Reference Log37
Part 1 - ESS Data: Start and End Times37
Part 2 - ESS Data: Start and End Positions42
Part 3 - DAT Acoustic Tapes46
Part 4 - Computer Acoustic Files50
Part 5 - VPR Acoustic Tapes and Bio-Optic Files55
INTRODUCTION
R/V Oceanus 334 was conducted as a U.S. GLOBEC process cruise to examine late autumn conditions in the Gulf of Maine relevant to ecosystem processes on Georges Bank. The major scientific objectives of the cruise included:
1. conducting broad-scale surveys to map the spatial distribution of Calanus finmarchicus and its major invertebrate predators in the deep basins of the Gulf of Maine,
2. conducting broad-scale surveys to map the spatial distribution of bio-optical properties in the Gulf of Maine.
Summaries of the two primary projects supported by this cruise are presented in sections following the cruise narrative.
R/V Oceanus 334 was planned as a broad-scale, bio-optical and bioacoustical survey cruise traversing the major basins of the Gulf of Maine. We had weather-based contingency plans for conducting this survey cruise in either a clockwise manner (Wilkinson Basin, Jordan Basin, Georges Basin) or the reverse. A grid of regularly spaced transect lines was laid out for the survey of each basin. In addition to the survey itself, time was allocated for daytime and nighttime MOCNESS tows as well as mid-day light stations.
CRUISE NARRATIVE
The R/V Oceanus departed Woods Hole on cruise OC-334 at 13:45 on 3 December 1998. Departure had been delayed two days getting the J-frame operational and devising a satisfactory launch and recovery plan for the BIOMAPER II (BIo-Optical Multiple-frequency Acoustical and Physical Environmental Recorder). After departure, we tested the deployment and recovery plan in Nantucket Sound. It worked better than expected, and we set out to Wilkinson Basin in the Gulf of Maine. A map of the cruise track and stations is presented in Figure 1.
We arrived at our first deployment site in Wilkinson Basin during the late evening of 3 December. The BIOMAPER II was deployed at 23:54 (BMP 2) and all systems were checked. (See Figure 2.) By 01:21 on 4 December, we were towyoing in Wilkinson Basin and collecting our first survey data on the cruise. All systems operated properly, and we continued towyoing until arriving at the mid-day light station at 12:17.
At 12:30, we began the first mid-day light station activities. The BIOMAPER II was towed at 10 m while these activities were conducted. The activities included deployment of a tethered, free-fall vertical profiling radiometer (SMPR 1) and a CTD cast (CTD 1). The CTD cast was completed at 13:41.

After completion of this station, we began surveying with the BIOMAPER II again, initiating the first towyo at 14:08. Towyoing was continued until 22:39, at which point the instrument was brought to a depth of 10 m. A nighttime MOCNESS tow (MOC 1) was conducted from 23:06 (4 December) until 00:40 (5 December). During the MOCNESS tow, the BIOMAPER II was collecting data at 10 m. Noise interfered with acoustic data collection during this MOCNESS tow. We are uncertain as to the origin of this noise, but it appeared to be related to switching the smaller Dynacon winch to the MOCNESS on and off.
Towyoing with the BIOMAPER II began again at 00:56 and continued throughout the night and next morning until we reached the site of our second mid-day light station at 11:04 on 5 December. During the 10 hours of towyoing, BIOMAPER II performed well with only occasional computer crashes that temporarily disrupted data acquisition.
At 11:15, we began the second mid-day light station activities. The BIOMAPER II was towed at 10 m while these activities were conducted. The activities included deployment of the vertical
profiling radiometer (SMPR 2) and several CTD casts (CTD 2,3,4). The additional CTD casts were the result of faulty electronics during the first two casts. The light station was completed at 13:40.
At 13:51, we began a daytime MOCNESS tow (MOC 2). The BIOMAPER II was collecting data at 10 m during this net tow and the noise problem that we experienced previously did not reoccur. The MOCNESS tow was completed at 15:25 and the BIOMAPER II was recovered at 16:10. This completed all operations in Wilkinson Basin and we began the 7.5 h transit to Jordan Basin.
We arrived at our first deployment site in Jordan Basin just before midnight on
5 December. The BIOMAPER II was redeployed to 10 m at 23:59 (BMP 3) to begin collecting data during the nighttime MOCNESS tow. At 00:10 on 6 December, we began the MOCNESS tow (MOC 3). It was completed at 01:31. The first transect line in Jordan Basin was begun at 01:40, and towyoing continued until 10:56 when we arrived at the next mid-day light station.
At 11:00, we began the third mid-day light station activities. The BIOMAPER II was towed at 10 m while these activities were conducted. The activities included deployment of the vertical profiling radiometer (SMPR 3) and a CTD cast (CTD 5). The light station was completed at 12:13.
At 12:55, we began a daytime MOCNESS tow (MOC 4). The BIOMAPER II was collecting data at 10 m during the net tow. This MOCNESS tow was completed at 14:29.
Towyoing with the BIOMAPER II began again at 14:48. This continued until 15:03, when the VPR stopped working. The BIOMAPER II was recovered at 17:52 to repair the VPR. The connector into the strobe appeared to have leaked. It was cleaned and greased, and the BIOMAPER II was redeployed at 19:26 (BMP 4).
Towyoing with the BIOMAPER II began again at 19:42 and continued until recovery at 08:40 on 7 December. The VPR and 43-kHz, upward-looking transducer stopped working during this period (02:50 and 04:54, respectively). After inspecting the VPR for several hours, it was decided to head into Provincetown to seek assistance from Scott Gallager of WHOI. The weather had finally taken a turn for the worse that afternoon and evening, so the decision to suspend operations and steam into Provincetown was an easy one to make.
We arrived in Provincetown at approximately noon on 8 December. Scott Gallager arrived on the ship at 13:50, and after much clever detective work, was able to get the VPR operational again. He left the ship at 17:00, and the ship steamed out of Provincetown toward Georges Basin at 20:00.
At 09:00 on 9 December, we deployed the BIOMAPER II at the starting point of our transect line in Franklin Basin (BMP 5), a southwest-to-northeast-oriented arm off the western end of Georges
Basin. Towyoing with the BIOMAPER II began at 09:01 and continued until arriving at the site of our afternoon MOCNESS tow at 14:43.
At 15:10, we began a daytime MOCNESS tow (MOC 5). The BIOMAPER II was collecting data at 10 m during the net tow. This MOCNESS tow was completed at 17:20, after dark. There was evidence of vertical migration during the net tow and these samples should not be viewed as representative of the daytime distribution of zooplankton.
Towyoing with the BIOMAPER II began again at 17:34. A transect line was conducted in the main portion of Georges Basin until we reached the cutoff point to the site on Georges Bank where we intended to deploy Scott Gallager's VPR profiler. The cutoff to this site was reached at approximately 01:00 on 10 December. From 01:00 until 08:56, a series of transects were run on and off Georges Bank in search of Peter Wiebe's elusive internal waves. At 08:56, just as we were preparing to deploy Gallager's VPR profiler, the BIOMAPER II telemetry system crashed. This was the first indication that the day was going to be a long one.
At 09:14, the VPR profiler was deployed. After it reached the air-sea interface, the
bridle lines supporting the profiler parted. The profiler was supposed to be lowered to its position near the sea floor and then acoustically released. Instead, it dropped to the bottom under its own weight. It could not be determined if the profiler landed in an upright position or not. It was decided that the best course of action was to leave the profiler in place until its planned March recovery.
The BIOMAPER II was recovered at 09:54, while we were still at the VPR profiler deployment site. At 10:15, we began mid-day light station activities at this site. These activities included a CTD cast (CTD 6) and deployment of the vertical profiling radiometer (SMPR 4). The light station was completed at 11:32.
We left the VPR profiler deployment site after the light station and steamed to GLOBEC Broad-Scale Station 29 in Georges Basin. At 13:24, we began a daytime MOCNESS tow (MOC 6) at this site. The net tow was completed at 15:22. After the MOCNESS tow was completed, a CTD cast was attempted. During deployment, the bail on the CTD rosette frame broke and the instrument was barely recovered. CTD use on the cruise came to a halt.
After several hours of troubleshooting, it was determined that a transceiver card in the winch's telemetry unit was the cause of the BIOMAPER II's telemetry system failure. Since we had no spare for this card, we were forced to switch telemetry from the electro- optic tow cable to a shorter piece of conventional cable. This shorter cable provided the communication link between the BIOMAPER II underwater and surface units. It was attached with tie wraps and tape to the electro-optic tow cable, which still provided the necessary towing strength. In this configuration, we could collect data, but could no longer towyo BIOMAPER II.
At 22:00, the BIOMAPER II was deployed at 6 m for surface-based operations along a 70-nautical mile, east-west transect line. The instrument was operated in this manner until 12:22 on 11 December. During this time period, a nighttime MOCNESS tow was conducted at 22:42 and the vertical profiling radiometer was deployed at 10:36. The MOCNESS was deployed near the eastern end of the transect from 22:42 until 00:27 on 11 December (MOC 7). The radiometer deployment was conducted near the western end of the transect from 10:36 until 11:35 (SPMR 5). With weather conditions rapidly deteriorating, we recovered the BIOMAPER II at 12:22.
With the BIOMAPER II on deck, we steamed to a site in Franklin Basin, near the northwest flank of Georges Bank, for an internal wave study led by Peter Wiebe. When we arrived at 17:24, the sea conditions were not suitable for deploying the instrument. Conditions improved overnight, and we were able to deploy the BIOMAPER II at 8:10 on 12 December. From this point until 17:57, we repeated a transect line on and off Georges Bank in search of internal waves generated as tidal currents flow from the Bank to the deeper waters of Franklin Basin. Finally, at 10:05, we recorded such waves cascading off the Bank just at the point in the tidal cycle when they are predicted.
At 18:01, we recovered the BIOMAPER II and began the steam to Woods Hole.
INDIVIDUAL REPORTS
1.0 Principal Project: Processes Regulating the Recruitment of Calanus finmarchicus from the Gulf of Maine to Georges Bank
Principal Investigators: C.H. Greene (Cornell), M.C. Benfield (LSU),
P.H. Wiebe (WHOI)
1.1 Introduction
Calanus finmarchicus dominates zooplankton secondary production on Georges Bank during spring. The Georges Bank Calanus population is derived from diapausing populations in the deep basins of the Gulf of Maine, the Slope Water, and the Scotian Shelf east of the Bank. It is hypothesized that processes that regulate the survivorship and supply of Calanus from diapausing populations in the deep basins of the Gulf of Maine determine this copepod species' annual production on Georges Bank. To test this hypothesis, research is being conducted 1.) to determine what processes regulate the abundance and survivorship of the diapausing Calanus populations in these deep basins, and 2.) to determine how physical and biological processes interact to seed Georges Bank with recruits from these basins each year.
Acoustic and video remote-sensing methods are used to survey diapausing populations of Calanus in Georges, Jordan, and Wilkinson Basins of the Gulf of Maine. Broad-scale survey cruises conducted in early and late autumn during 1998 and 1999 will be used to estimate survivorship of the diapausing Calanus and the scale-dependent spatial coupling of Calanus and its principal invertebrate predators - the euphausiid, Meganyctiphanes norvegica, and the siphonophore, Nanomia cara.
Results from field studies and feeding experiments will focus on quantifying the relative contribution of predation to the overall mortality of the diapausing Calanus population. The results from field studies also will be used as input to coupled physical-biological models to examine how physical transport processes in the Gulf of Maine interact with Calanus seasonal and diel vertical migration behaviors to seed Georges Bank with new recruits during winter.
1.2 Methods
1.2.1 Broad-scale survey
A broad-scale survey of the Gulf of Maine deep basins was conducted from December 3 - 13, 1998. The survey track included multiple transect lines in Wilkinson, Jordan, and Georges Basins (Figure 1). The survey was conducted using a high-speed, deep-towed system, the BIOMAPER II (BIo-Optical Multiple-frequency Acoustical and Physical Environmental Recorder), configured with multiple-frequency (43 kHz, 120 kHz, 200 kHz, 420 kHz, and 1 MHz) acoustics, a video plankton recorder (VPR), and several bio-optical sensors (down- and upwelling spectral radiometers, spectrally matched attenuation and absorption meters). The acoustic frequencies were chosen to bracket the transition from the Rayleigh to geometric scattering regions for the target species under investigation. A down- and up-looking transducer operated at each frequency to provide more complete coverage of the water column when the instrument was towyoed. Echo integration was conducted at 12-second intervals to provide volume-backscattering data at all five frequencies. Split-beam data was collected at the four lower frequencies. All raw acoustic data were recorded on digital audio tape, while the processing for echo integration was carried out in real time.
1.2.1.1 Multi-frequency acoustics
The BIOMAPER II system consisted of the towed body, a 1000 m piece of 0.68" electro-optical cable on the ship=s Dynacon traction trawl winch, and a standard 20' shipping Van converted into a sea-going laboratory. For this cruise, BIOMAPER II was carrying the HTI acoustic system with 5 pairs of transducers (43, 120 200, 420, and 1000 kHz -up and down looking), a Video Plankton Recorder (VPR), an Environmental Sensing System (pressure, temperature, conductivity, fluorescence, downwelling light, beam transmission), and the Sosik Bio-optical package (2 ac-9's, and upwelling and downwelling Spectral Radiometers). Two subsurface and six van based PC microcomputers were used in the real-time data acquisition and data processing (see figure from OCEANUS cruise 332 report).
Launch and Recovery of BIOMAPER II
On OCEANUS Cruise 334, BIOMAPER II had to be launched and recovered by the ship=s crane located on the O-1 deck, and towed from a newly reinforced J-frame located on the main deck=s starboard quarter. The towing wire (0.68 electo-optical wire with three copper conductors and 3 glass fibers, and a breaking strength of 46,000 lbs) was spooled onto the drum of the ship=s Dynacon traction winch located in a room underneath the stern portion of the main deck. At the start of the cruise, the Bosun, Jeff Stolp, organized members of the scientific party so that there was a person for each job associated with operation and that person always was responsible for doing that job. Joe Warren and Karen Fisher ran the air tuggers, Andy Girard controlled the mechanical quick release. Karl Fisher kept lines clear and installed or removed straps holding BIOMAPER II on the deck, Peter Wiebe guided the trawl cable and worked the J-frame. A crew member ran the winch and another ran the crane.
Launch and recovery was done with the ship steaming ahead at about 1.5 to 2.0 kts. BIOMAPER II was located on the starboard deck aft of the CTD. It was secured to the deck with heaviest of the aircraft tie-down rachet straps, two per side. A pair of air tuggers were positioned inboard and ahead and behind of BIOMAPER II. The air tuggers reels were loaded with 3/8" Spectra line with a closing snap hook attached to the end. A pair of deck cleats were bolted to the deck near by the tuggers and a bit outboard of them. Screwed into the deck next to the cleats and still further outboard was an eye-bolt. To prepare to launch the fish, two lines were attached to the eye-bolts on the deck. These lines were lead to the stainless rail along the top inboard side of the fish, passed over the rail, and then down to the cleat where they were figure-8ed. A lifting strap was put into place around the lift point on BIOMAPER II=s towing bail and this strap was attached to mechanical quick release mechanism. While the lines and lifting strap were being put into place, the Bosun was directing the crane to move to a position near the J-frame and then the head-ache ball and jib cable were lowered to where he could grab a hold of it. The Bosun walked the head-ache ball forward as the crane was then maneuvered to a position just outboard of BIOMAPER II. The quick-release-mechanism=s pear-ring was snapped into the head-ache ball hook and the slack taken out of the lifting assembly by hauling in on the crane=s jib cable. With the 0.68" electro-optical cable being held inboard of the fish (to prevent it from coming in contact with the transmissometer or tail assembly and light sensors), the jib cable was hauled up while the restraining lines were held tight. The fish was lifted off the deck, but held inboard of the ship=s railing by the restraining lines until it was high enough to clear the railing. Then the lines were eased as the crane boom was lowered to move the fish outboard while the jib cable continued to be raised. Once the fish was well clear of the railing, the jib cable was lowered fairly quickly to get the fish into the water and under the sea surface. The restraining lines were cleared as the fish entered the water.
The next step was to move the fish aft to a position outboard of the J-frame where the transfer of the load from the crane to the towing cable took place. This was done by slowly moving the crane boom aft and raising it upward. During this period, the slack towing cable was pulled to the inboard portion of the towing block hanging from the J-frame and a person held the wire to put tension on it as it was pulled in by the Dynacon trawl winch. Once the slack was out of the towing wire and the J-frame moved to its down and outboard position, the line to the quick release mechanism was pulled and the weight of the fish transferred to the towing cable. BIOMAPER II was now ready to be towyoed.
Recovery was largely the reverse of this process except that the tugger lines were used instead of free lines. The tugger lines were spooled out, and the closing hooks and line were led to the back portion of the deck behind the J-frame. A square SS bar welded to the back side of each closing hook was slipped into one end of a long aluminum pole with a special insert to allow the bar to slide freely in or out. The lifting strap, also equipped with a snap hook and bar for pole deployment, was readied. The Bosun motioned for the crane whip to be lowered from the crane which was positioned so that the head-ache ball would drop to the rail just aft of the J-frame. He then motioned to the winch operator to haul in on the trawl cable to bring BIOMAPER II to the surface so the top of the fish was just in and out of the water as waves moved passed. Andy Girard, holding a line tied to the lifting strap tight against the aluminum pole, extended the lifting strap snap hook down to the fish and hooked onto the lifting bail. Then the other end of the lifting strap was snapped into the head-ache ball hook and the Bosun motioned for the crane operator to take out some of the slack in the lifting strap, but not enough to transfer the weight of the fish to the crane. Joe Warren and Karen Fisher then moved up to the rail with their closing hooks on poles and extended them down to hook onto the SS rails along the top port side of the fish. Karen hooked on the forward section and Joe hooked on the aft section. Finally, Andy re-used one of the poles fitted with a third closing hook tied to a strong line to hook into the aft section as well. With the lines in place, the Bosun motioned for the trawl winch operator to begin paying out cable while also having the crane move outboard away from the ship and to take up slack with the whip, thus transferring the weight of the fish to the crane. With a person putting tension on the trawl cable to keep it from unwinding and catching up in the winch room, enough cable was spooled off the trawl winch to allow the fish to be moved forward and landed on the deck. About the same time, the J-frame was moved to the upright inboard position, making the rail clear for people to move along it. Karl Fisher made sure the tugger lines which were still slack were kept clear of any potential hangups; Andy kept his tail line in hand; and Peter Wiebe made sure the trawl cable was clear of the transmissometer and tail assembly. The fish was then moved forward by slewing the crane forward. Once the fish was clear of the J-frame and abeam the deck area where the fish would be landed, the tugger lines were tightened, but not severely. The Bosun motioned for the crane to lift the fish out of the water and as it rose up out of the water the tugger lines were tightened and the aft line was looped around a cleat to hold the tail from hobby horsing up and down. Slowly and steadily, the fish was raised above the rail and brought inboard with the crane while the tugger lines were kept taut. Once inboard, the crane whip was lowered while keeping the tugger lines tight. Adjustments to the landing location were made by slight changes in the tightness of the tugger lines. The tugger lines were kept taut after the fish was landed while the air craft straps were installed and tightened to hold the fish securely on the deck starting first with the inboard straps and then the outboard straps. A thick piece of white plastic sheeting was bolted into place over the up-looking transducers as a protective cover. Then the tugger lines were released and wound up, and the other hand lines were coiled and put away.
1.2.1.2 Video Plankton Recorder
The VPR mounted on BIOMAPER sampled a volume of .234 liters min-1. All video images were recorded on S-VHS tape for post-cruise processing. GPS data were synchronized with the VPR signal and stamped on the video time code.
1.2.2 MOCNESS Zooplankton Samples
Two MOCNESS profiles were done in each of Wilkinson and Jordan Basins; three profiles were done in Georges Basin. As much as possible, one day and one night tow was completed in each Basin. The tow depth was generally to within 10 m of the bottom. Tows were designed to sample 25 m depth strata near the bottom and near the surface of the tow.
The MOCNESS samples were used for several projects, which required sample splitting and preservation using a variety of methods. One-half splits were preserved in 10% buffered formalin for silhouette analysis to ground-truth the bio-acoustic measurements. One-quarter splits were made up to 600 ml, and 180 ml were removed and preserved in 5% formalin for C.B. Miller=s (OSU) studies of copepod life history. The remainder of the 600 ml were placed on ice for later sorting of target species for molecular studies by A. Bucklin (UNH). The final one-quarter split was sieved and spooned into plastic bags and frozen for stable isotope analyses by K. Fisher (Cornell), and molecular analyses by P. Hassett (Ohio Univ.) and A. Bucklin (UNH).
1.3 Results
1.3.1 Broad-scale survey
1.3.1.1 Multi-frequency acoustics
The multi-frequency acoustics, bio-optical (including VPR), and physical oceanographic data were collected during seven deployments of BIOMAPER II throughout the Gulf of Maine (Table 1). Deployment 1 was a test of the gear in Nantucket Sound; deployment 2 was in Wilkinson Basin; deployments 3 and 4 were in Jordan Basin, deployments 5 and 6 were in Georges Basin, and deployment 7 was on the Northwest portion of Georges Bank and Franklin Basin. On this cruise with the deployment of BIOMAPER II from a J-frame mounted on the starboard quarter of the deck, the actual transect lines followed the planned lines with few deviations. The total trackline distance along which data were collected was 930 km (502 nm - Table 1). During the deployments, data were collected in a down-looking mode with BIOMAPER II towed just below the surface, in a down and up-looking mode while towed horizontally at some sub-surface depth, or towyoed from the surface to within 10 to 30 m of the ocean bottom. As described above, during some periods of BIOMAPER II data acquisition, MOCNESS tows were taken to collect planktonic animals.
Table 1.Distance of BIOMAPER II Trackline Sections on R/V OCEANUS Cruise334 (5-13 December 1998).
DeploymentYear-day Timekmnm Area
StartEnd
1337.70694337.72361--Nantucket Sound
2337.99583339.67361287.6155.3Wilkinson Basin
3339.99930340.74444128.169.2Jordan Basin
4340.80972341.36111121.365.5Jordan Basin
5343.37569344.4125198.1107.0Georges Basin
6344.91666345.5152795.451.5Georges Basin
7345.34027345.7518.39.9Georges Basin
8
346.35346.7478280.843.6NW Georges BankTotal929.6502.0
The instrument systems on BIOMAPER II did not work well on this cruise. There were significant problems with the VPR and the Bio-Optical subsystems, and the up-looking 43 kHz transducer failed midway through the cruise while the towed body was about 190 m below the surface during a towyo in Jordan Basin. The VPR problem was caused by an electronic failure of the strobe light system. After the first failure, the ship steamed to Provincetown, Ma where Scott Gallager came out to the ship to trouble shoot the system and install a replacement part. It failed a second time while work was ongoing in Georges Basin, but no attempt was made to repair it at sea.
The software to acquire the data, which was provided by HTI, enabled the simultaneous acquisition of data on five frequencies each with two transducers (one up-looking and one down-looking). The range of the 0.5 meter depth strata allocated for each transducer was dependent on frequency with the lowest frequencies given the longest range and highest frequency the shortest range i.e. 43 kHz = 200 m, 120 kHz = 200 m, 200 kHz = 149, 420 kHz = 100, 1000 kHz = 35). Data were collected in 12 second intervals (averages). A ping cycle for all frequencies and transducers took about 2.5 seconds with a firing sequence of down-looking 43, 120, 200, 420, 1000 kHz and then up-looking 43, 120, 200, 420, 1000 kHz. Target Strength measurements were also collected on the lower 4 frequencies out to 20 meters from the surface of the split beam transducers. In addition, the raw data for each ping were recorded on DAT tape for postprocessing of either target strengths or echo-integrations on a different time averaging basis. Each tape recorded two gigabytes of data and a total of 57 tapes were recorded during the cruise.
Post-processing software was used to combine the up-looking and down-looking data for a given frequency and then display the data as color enhanced echogram. On this cruise, a new MATLAB version of the program to translate HTI-DES formatted acoustic data to a MATLAB compatible format (the Wiebe format) that was written by Andy Pershing was used as the first step in post-processing the acoustic data. Code, developed by Joe Warren, was used to create visual images of these data. While at sea, new software was written principally by Andy Pershing to visualize the environmental data (temperature, salinity, fluorescence, and transmissivity). As a result, plots of the volume backscattering in the water column and associated environmental properties were available shortly after a BIOMAPER II section had been completed.
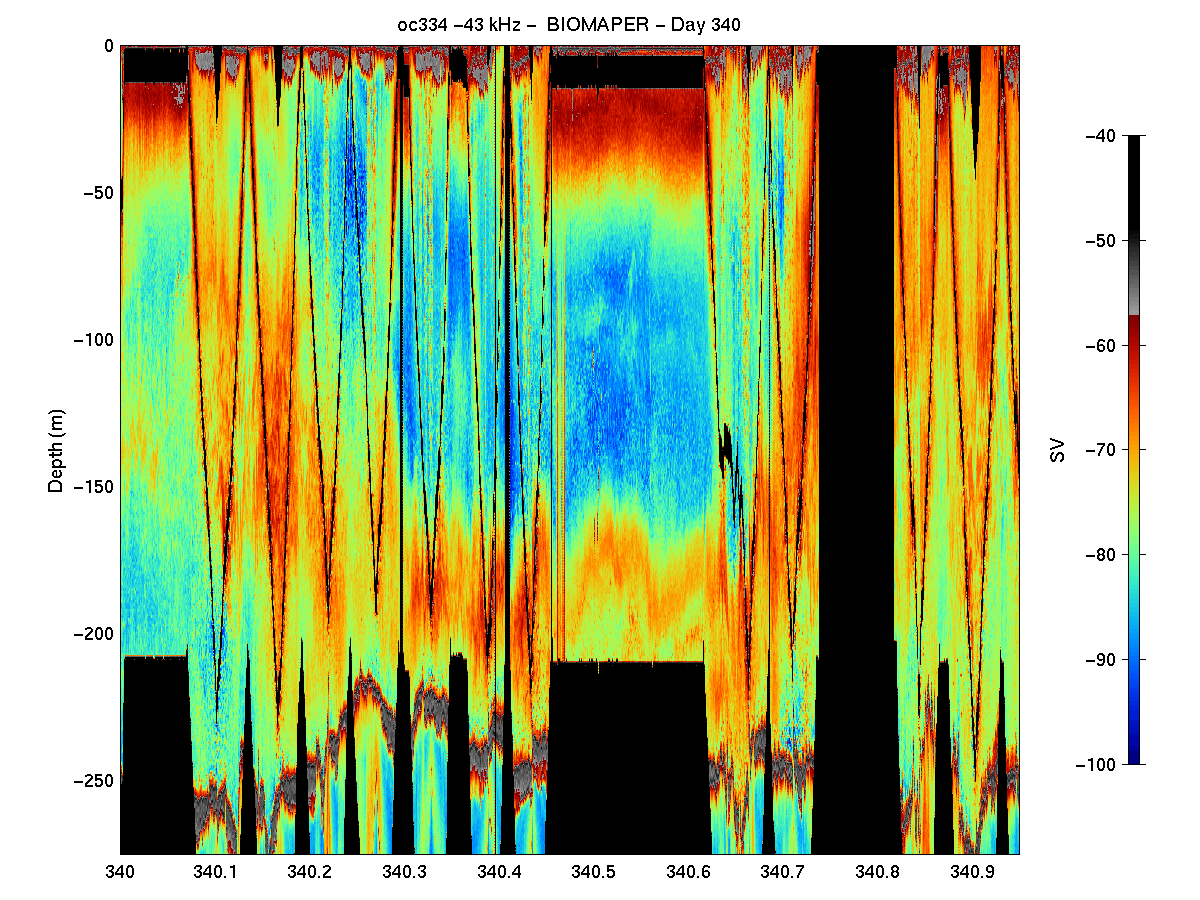
Some Results: There are several general observations that result from an initial look at the post-processed acoustic data. Highest volume backscattering throughout the water column was present in the upper 25 to 50 meters both day and night in all three Gulf of Maine Basins. Diel vertical migration was evident in the echograms from all three basins with somewhat lower volume backscattering in the surface waters during the day and higher levels in this depth interval at night (Figure 3). During the same period, increased scattering occurred at mid-depth
levels (i.e. >100 meters) which became much weaker at night, but not absent. In fact, in Wilkinson and Jordan basins, there was substantial subsurface volume backscattering layers at night.
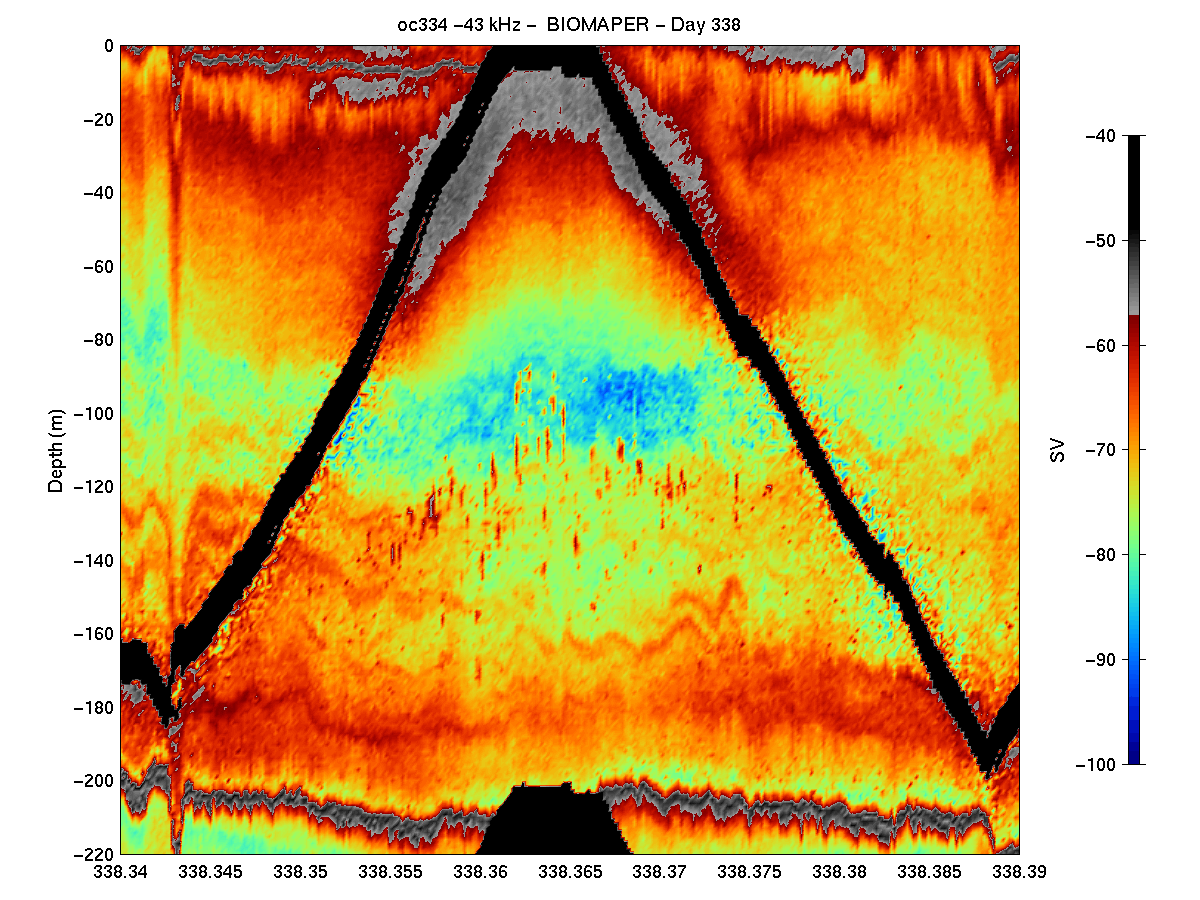
There were other subtle basin to basin differences in the acoustic structure as well. A distinct layer was present near the bottom in both Wilkinson and Jordan Basins which did not show up as well in Georges Basin. Also in Wilkinson Basin there were some isolated, but quite strong targets (perhaps small fish schools) which occurred between 100 and 150 m depth during the
daytime which were not evident in the other two basins (Figure 4). Interpretation of these trends in terms of the biological entities present must await the analysis of the VPR video tapes and the MOCNESS samples.
1.3.1.2 Video plankton recorder (VPR)

BIOMAPER II was equipped with a single-camera VPR mounted on the front of the upper surface of the vehicle with the camera (starboard) and strobe (port) facing inward (Figure 5). The separation distance between the camera and strobe transparent ports was approximately 28.5 cm. The camera imaged a volume of 4.6 ml with dimensions of 17 mm wide x 13.0 mm tall x 21 mm deep. The center of the volume was located approximately 12 cm from the camera pressure port. The actual volume that was detected by the frame grabber has a more limited height because the section of the video image where the time-code is displayed is not included. Thus, the effective volume was 3.9 ml per field with dimensions of 17 mm wide by 11 mm tall by 21 mm deep. The strobe was not equipped with a red filter and it illuminated targets with white light that flashed at 60 Hz synchronized to the camera with a 20 ms pulse length. Light from the strobe was aimed obliquely relative to the camera so that dark field illumination of targets was achieved. See R/V Oceanus 332 cruise report for further details.
1.3.2. MOCNESS Zooplankton Samples
Based on inspection of the MOCNESS samples, it appeared that Wilkinson Basin had
the highest abundances of Calanus finmarchicus and other copepods. However, the assemblage was mixed, and there was no evidence of the dense diapausing populations of C. finmarchicus, that might be expected at this time of year. All three Basins had sparse populations of very small juvenile C. finmarchicus (stages CIV and CV) occurring 35 - 60 m off the bottom. This finding is consistent with Miller's findings on the June broad-scale survey cruise. The most notable components of the zooplankton assemblage of the Basins was the overwhelming abundance of pteropods (Limacina sp.) at depths dependent upon the time of day. It appeared that the pteropods were feeding upon dense surface masses of filamentous diatoms (also somewhat unusual for this time of year) at night and returning to depth during the day. In turn, the pteropods seemed to have attracted many predatory naked pteropods, Clione sp.
The relative abundances of important taxa were characterized by swirling the quart jar with the one-half split in formalin. This qualitative assessment revealed clear vertical stratification of the zooplankton in all three Basins, and clear differences between the three Basins in terms of the predominant species found. Notable differences included the abundance of the planktonic polychaete, Tomopteris sp., in Jordan Basin and thick concentrations of the siphonophore, Nanomi cara, in Georges Basin.
2.0 Ancillary Project: Spatio-Temporal Variability of Bio-Optical Properties in the Gulf of Maine
Principal Investigator: H.M. Sosik (WHOI)
2.1. Introduction
The optical properties of coastal waters are influenced by a variety of materials and processes. Inherent optical properties (e.g., absorption and scattering coefficients) depend strongly on local production of phytoplankton and other optically-active particulates, but are also influenced by advection of constituents, such as dissolved organic material of terrigenous origin, and other processes such as resuspension of bottom sediments. Our objectives are to determine important sources and scales of optical variability by observing distributions of inherent and apparent optical properties in both space and time. Separately assessing particulate and dissolved material properties and making spectral measurements (which can be used to characterize broad classes of material) will allow this to be accomplished. The work conducted on this cruise is part of a larger program supported by the Office of Naval Research and which includes measurements made from a vertically-profiling mooring, a towed vehicle (BIOMAPER), earth-orbiting satellites (SeaWiFS, AVHRR, etc.), and research vessels conducting conventional station surveys.
2.2. Methods
Several bio-optical sensors were deployed on BIOMAPER II. These included two ac-9 dual-path spectral absorption and attenuation meters (Wet Labs, Inc.), as well as a spectral downwelling irradiance sensor (OCI-200 series, Satlantic, Inc.) and a spectral upwelling radiance sensor (OCR-200 series, Satlantic, Inc.). One of the ac-9's measured whole-water properties and the other was configured with an in-line 0.2 mm water filter to assess absorption by dissolved material; particulate absorption will be determined by difference between total and dissolved. The instruments were configured with matching wavelength bands selected to cover the visible spectrum and to reflect those on the SeaWiFS ocean color sensor [ac-9's: 412, 440, 488, 510, 532, 555, 650, 676, 715 nm; radiometers: 412, 443, 490, 510, 555, 665, 683 nm]. The data acquisition system (designed and assembled at WHOI) for these instruments is based on a subsurface PC-104 and includes 2 serial ports, a 16-channel/16-bit A/D converter, an 8 MB flash disk, and an ethernet adapter for communication with the BIOMAPER II Lantastic network. Through network access, this system allows storage of data files on a desktop PC aboard the ship. In addition to the measurements made from BIOMAPER II, continuous logging of surface spectral irradiance was also carried out with a spectral downwelling irradiance sensor (MVDS, Satlantic, Inc.) connected to the same PC.
Sampling conducted on the cruise also included water collection and measurements of downwelling irradiance and upwelling radiance with a tethered, free-fall vertical profiling radiometer (SPMR/SMSR system, Satlantic, Inc.) deployed away from the ship. This work was conducted at mid-day on each day of the cruise. Water samples were collected for pigment analysis and for estimation of light absorption by particulate and dissolved material. Pigment samples were collected on Whatman GF/F filters and extracted in 90% acetone for fluorometric estimation of chlorophyll a and phaeopigment concentrations. Spectrophotometric determinations of particle absorption were made on freshly filtered samples (GF/F filters) before and after extraction of phytoplankton pigments with methanol, and dissolved absorption was measured on 0.2 mm filtrate in 10 cm cuvettes. These spectra were acquired with 1 nm resolution between 300 and 800 nm. At all stations water samples also were collected and frozen or preserved for several other types of post-cruise analysis, including determination of macronutrient concentrations, flow cytometric measurement of individual particle optical properties, and microscopic identification of phytoplankton.
The vertically profiling radiometer deployed at each of the mid-day CTD stations has the same spectral bands as the optical heads on BIOMAPER II. The system also includes a subsurface reference sensor (spectral downwelling irradiance at 30-cm depth). Vertical profiles were conducted within 2 hours of local apparent noon, immediately prior to or immediately after the CTD/water sampling casts.
Underway measurements with a Fast Repetition Rate Fluorometer (FastTracka FRRF, Chelsea Instruments) also were made in flow-through mode using the ship's uncontaminated seawater for most of the cruise duration. This instrument allows the photosynthetic physiology of phytoplankton to be measured rapidly and non-invasively. Dark-adapted measurements of variable fluorescence yield, the functional absorption cross-section for photosystem II reaction centers, and the turnover time for electron transport were made.
A Coulter Multisizer was used to make measurements of particle size distribution on water sampled with the CTD and the underway flow-through system. The counter was fitted with a 50-mm aperture such that reliable distributions were obtained in the size range of 1 to 30 mm.
2.3. Results
Optical data were collected from the BIOMAPER II during the deep basin surveys. Initial processing suggests that operation in towyo mode proved successful in mapping distributions of optical properties. Data collected show spatial variations in scattering and absorption coefficients associated with water column structure. Highest values were found near the bottom and in conjunction with phytoplankton patches in the upper 50 m.
Approximately 35 water samples were analyzed for pigment, high spectral resolution absorption and particle size distribution. Water was collected from 7 or 8 depths selected from throughout the water column on 4 CTD/rosette casts and from two days of surface sampling collection. During the surface sampling collection days, samples were coordinated with the closest BIOMAPER upcast so that BIOMAPER was at the surface. Pigment concentrations and particle absorption coefficients exhibited subsurface maxima (several mg l-1) at a depth of approximately 30 m, with magnitudes falling off rapidly below 50 m. Absorption by non-phytoplankton particulates was very low in the surface waters.
The free-fall radiometer was successfully deployed at 5 stations with 2-3 replicate vertical profiles conducted during each deployment. Casts with the radiometer covers in place were also carried out to allow correction for temperature dependence of dark values. These data will be analyzed to determine vertical profiles of diffuse attenuation coefficients and remote-sensing reflectance.
Throughout the entire cruise data were collected with the FRR fluorometer. Preliminary results show daily variations in photosystem II efficiency, with inhibition evident just after local noon on a sunny day and absent on a cloudy day.
CRUISE PARTICIPANTS
Scientific Personnel
1.Charles Greene - Cornell University (Chief Scientist)
2. Louise McGarry - Cornell University
3. Karen Fisher - Cornell University
4. Andrew Pershing - Cornell University
5. Andone Lavery - Cornell University
6. Mark Benfield - Louisiana State University
7. Peter Wiebe - Woods Hole Oceanographic Institution
8. Ann Canaday - Woods Hole Oceanographic Institution
9. Rebecca Green - Woods Hole Oceanographic Institution
10. Joseph Warren - Woods Hole Oceanographic Institution
11. Andrew Girard - Woods Hole Oceanographic Institution
12. Laura Stein - Woods Hole Oceanographic Institution
13. Ann Bucklin - University of New Hampshire
14. Rod Berens - unaffiliated
15. Karl Fisher - unaffiliated
Ship's Officers and Crew
1. Lawrence Bearse (Master)
2. Courtenay Barber III
3.Anthony Diego Mello
4. Jeffrey Stolp
5. Horace Medeiros
6. James Ryder
7. Patrick Pike
8. Patrick Mone
9. John Kevin Kay
10. Alberto Collasius, Jr.
11. Torri Corbett
12. Brett Maloney
APPENDICES
Appendix 1. Event Log21Appendix 2. Acoustics Log23
Appendix 3. Cross Reference Log37